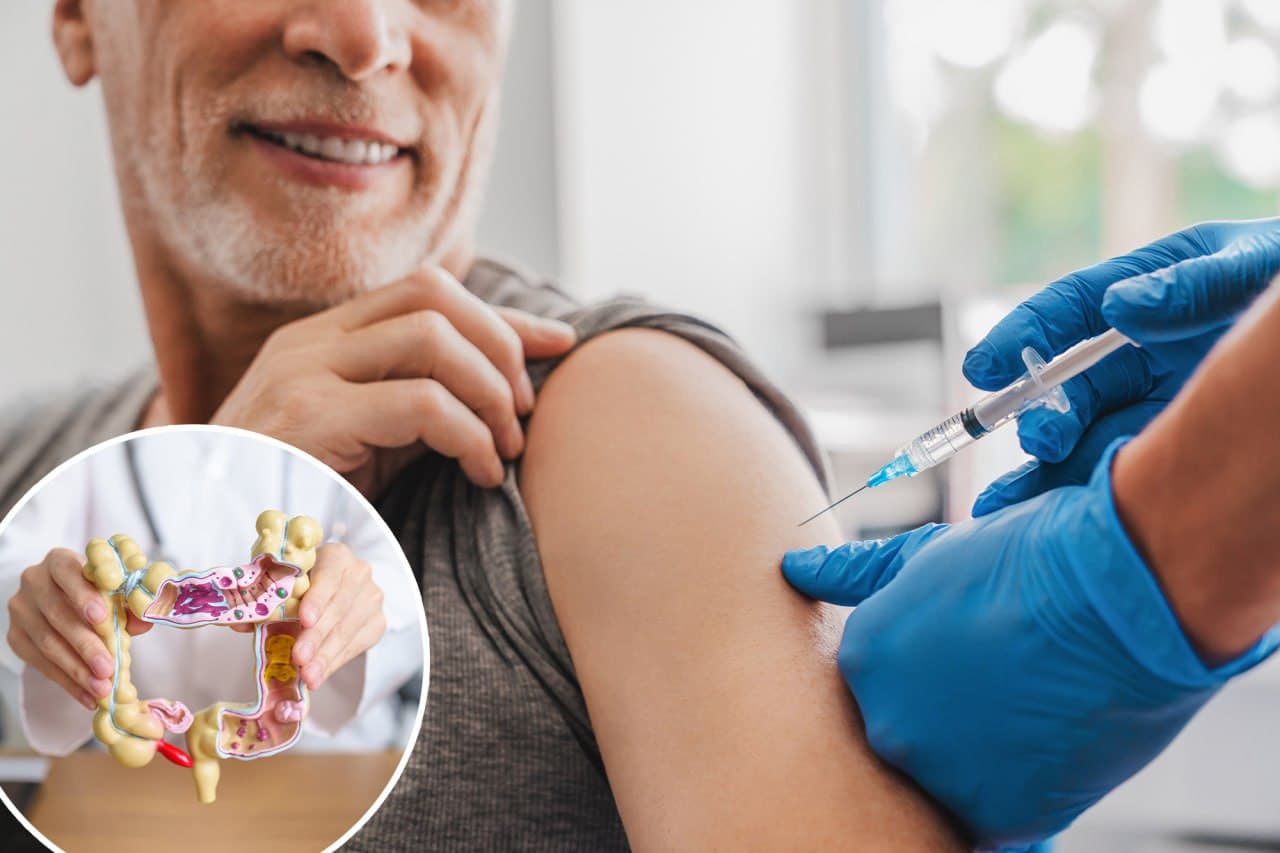
A new experimental cancer vaccine, ELI-002 2P, has shown promising results in preventing recurrences of pancreatic and intestinal cancers, according to a recent phase 1 clinical trial involving 25 patients. The vaccine successfully activated an immune response against KRAS gene mutations, which are responsible for up to 90% of pancreatic cancer cases and half of intestinal cancer cases.
In the trial, 21 out of 25 patients exhibited a strong immune response, generating KRAS-specific T-cells that extended the duration without cancer recurrences. Notably, in six patients—three with intestinal cancer and three with pancreatic cancer—all disease biomarkers vanished.
Patients demonstrating the most robust immune responses remained cancer-free for nearly 20 months post-vaccination. Developed by Elicio Therapeutics, ELI-002 is a “ready-to-use” solution, which eliminates the need for individual customization, thereby reducing costs and complexity.
The emergence of this vaccine offers significant hope for patients at risk of recurrence, marking a potential breakthrough in cancer prevention strategies. As researchers continue to monitor outcomes, the medical community watches closely for future developments in this area of cancer treatment. For more insights into related coverage, check out recent developments in other significant health issues.






![[Video] Gunfire between Iraqi security forces and Sadr militias in Baghdad](/_next/image?url=%2Fapi%2Fimage%2Fthumbnails%2Fthumbnail-1768343508874-4redb-thumbnail.jpg&w=3840&q=75)
